

A pluripotent stem cell is an undifferentiated cell that has the potential to divide and differentiate into any of the 220 cell-types of the human body. Stem Cell Therapy (SCT), is the utilization of these cells in order to assist the body in the healing and regeneration of its existing cells.
Depending on the conditions, pluripotent stem cells can be delivered through the blood stream or directly to the organ that is in need of treatment. Within 30 minutes of their delivery, the body starts to repair its damaged organs and the healing process can be observed up to 10 days after the injection. Several injections might be needed depending on the severity of the conditions and the expected results.
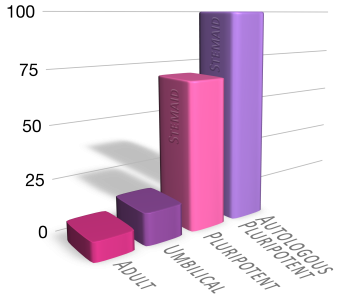
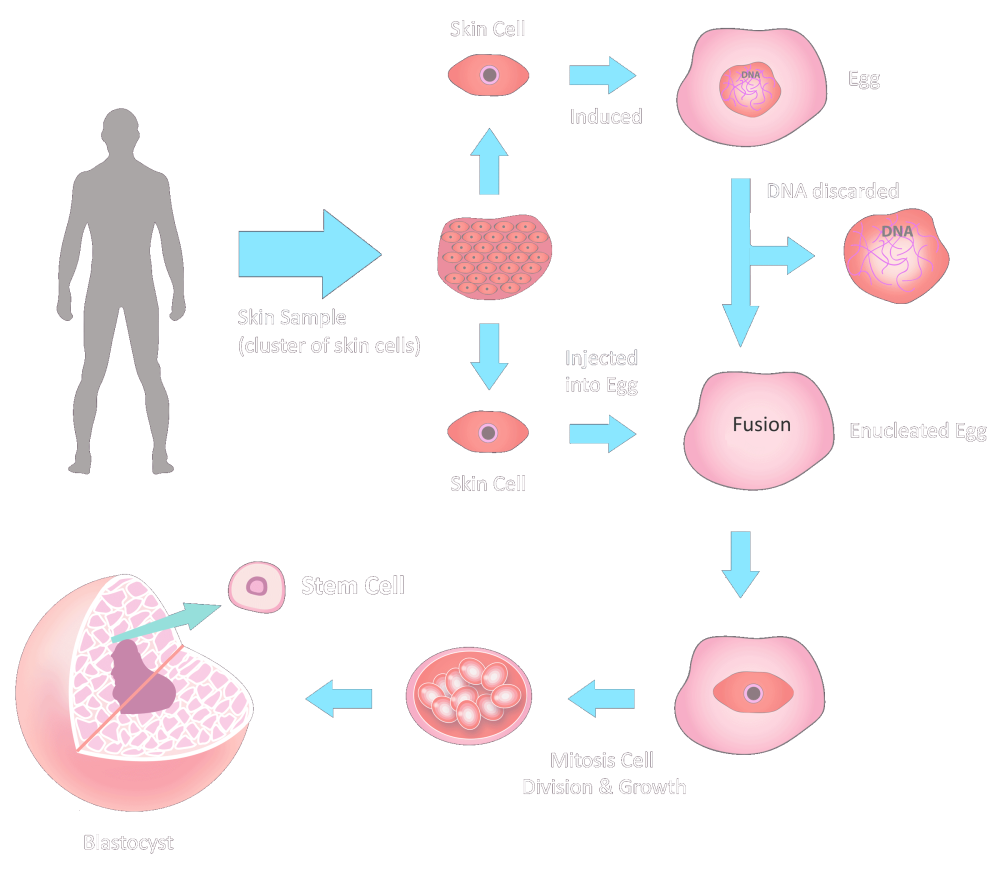
Traditionally, these cells were derived from donated blastocyts, which raised ethical and practical issues. Stemaid™ has developed a novel method to reprogram adult skin cells into pluripotent stem cells, without requiring any embryonic material. This means that we can produce stem cell lines that are customized for each patient, using their own skin cells. These are called autologous pluripotent stem cells.
Stemaid™ is the first company in the world to have produced pluripotent stem cells for therapeutic use. With over a decade of experience, you can trust that our stem cells are safe and reliable.
Company established and dedicated research begins.

Clinical trials begin.

Commencement of stem-cell distribution
Commence production of skin-cell-derived embryonic stem-cells.
Whether you're a patient seeking stem cell therapy or a doctor seeking stem cells for your patients, please feel free to get in touch with us.
OR
Call or Text/SMS to 1-520-STEMAID